The fashion industry generates trillions of dollars annually by selling the idea that clothing is simply fabric shaped into style, a neutral and passive material that sits against the body without consequence. The chemical reality of how modern garments are produced, treated, dyed, finished, and preserved tells a profoundly different story that the industry has very little financial incentive to communicate clearly to consumers. Skin is the body’s largest organ and its most constant point of contact with the textile environment and the compounds transferred from fabric to skin over hours of daily wear accumulate in ways that conventional toxicology has only recently begun to map with precision. The following forty-five mechanisms are ordered from the most broadly documented and widely experienced down to the more specific and technically obscure pathways through which the clothing industry’s chemical dependencies quietly reach the body.
Formaldehyde Finishing

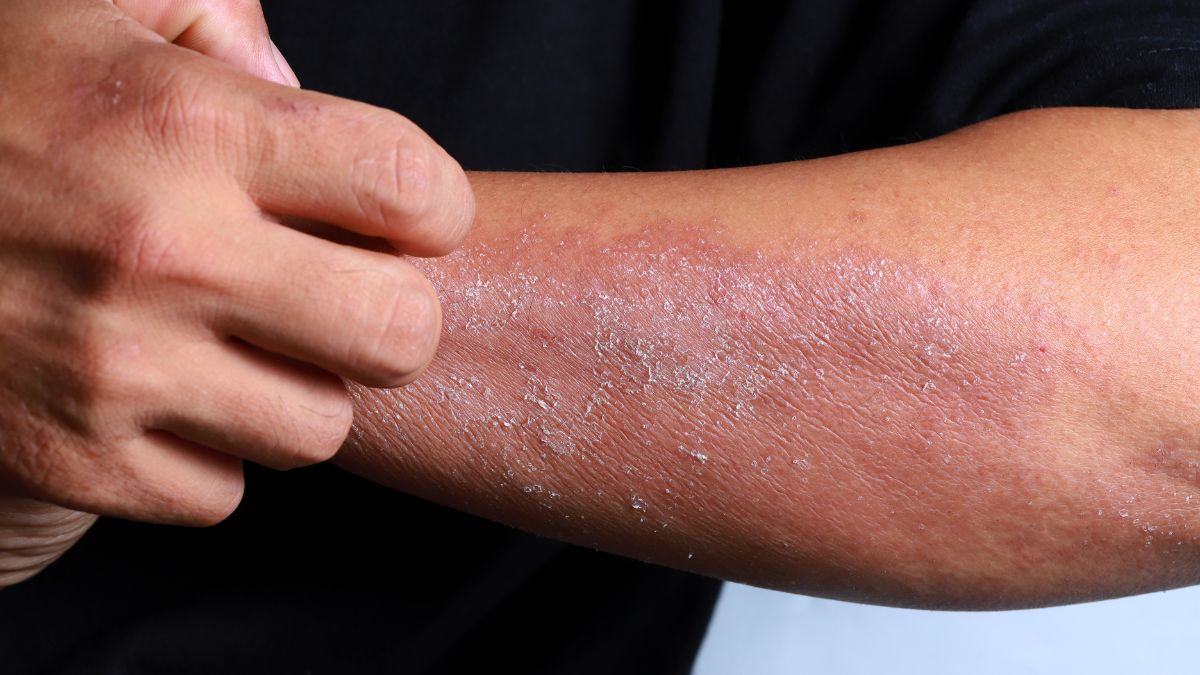
Formaldehyde resins are applied to fabric during manufacturing as a wrinkle-resistance and anti-shrink treatment and remain present in garments at measurable concentrations that transfer directly to skin through contact and perspiration. The compound is a documented human carcinogen at sufficient exposure levels and a potent contact allergen that produces dermatitis in sensitive individuals at concentrations well below those associated with carcinogenic risk. Regulatory limits for formaldehyde in clothing vary dramatically between jurisdictions with some major markets permitting concentrations that others have classified as unsafe for skin contact. Garments labeled as permanent press, wrinkle-free, or easy care carry the highest likelihood of formaldehyde resin treatment as these finishes are chemically dependent on the compound’s cross-linking properties. The concentration present in a new garment diminishes with washing but multiple studies have documented residual levels after repeated laundering that remain above threshold concentrations for dermatitis in susceptible individuals.
Azo Dyes

Azo dyes represent the largest single class of synthetic colorants used in textile production and certain azo compounds can cleave under the reductive conditions created by skin bacteria and perspiration to release aromatic amines classified as carcinogenic by international regulatory bodies. The release of these amines is not a manufacturing defect but a predictable chemical reaction that occurs when specific azo structures encounter the biological environment at the skin surface. Twenty-two specific aromatic amines released through azo dye cleavage are restricted or banned in the European Union while remaining in common use in garments manufactured in and imported from less regulated markets. Brightly colored garments and those with deep saturated hues carry statistically higher azo dye loads than pastel or naturally tinted textiles. The carcinogenic aromatic amines released through this mechanism are absorbed transdermally and accumulate in body tissue in patterns documented by biomonitoring studies.
PFAS Coatings

Per- and polyfluoroalkyl substances applied to clothing as water-repellent, stain-resistant, and soil-release finishes represent one of the most extensively documented categories of toxic textile contamination due to their extreme environmental and biological persistence. These compounds transfer from treated fabric to skin through friction and perspiration and accumulate in blood serum, liver tissue, and thyroid tissue in ways that have been associated with endocrine disruption, immune suppression, thyroid disease, and certain cancers in epidemiological research. The category includes thousands of distinct chemical structures and the regulatory phase-out of earlier long-chain PFAS compounds has been followed by their replacement with shorter-chain variants whose safety profiles are increasingly being questioned by independent researchers. Outdoor and activewear categories carry the highest PFAS loads given the performance expectations of these garments but the compounds appear across apparel categories far more broadly than most consumers appreciate. Blood serum PFAS levels in the general population correlate significantly with clothing and textile exposure in addition to food packaging and cookware contact.
Pesticide Residues

Conventionally grown cotton is among the most pesticide-intensive agricultural crops in the world and the residues of organophosphate, organochlorine, and pyrethroid pesticides applied during cultivation persist through fiber processing, spinning, and weaving into the finished garment at concentrations measurable by sensitive analytical methods. These residues transfer to skin during wear particularly in conditions of perspiration and heat that increase dermal absorption rates. Organophosphate pesticide residues specifically have been associated with nervous system effects at chronic low-level exposures and their presence in skin-contact textiles represents a route of exposure distinct from dietary or environmental pathways. The organic cotton certification process requires pesticide-free cultivation but certification standards are inconsistently enforced across producing regions and the premium commanded by certified organic cotton creates economic incentive for fraudulent labeling in opaque supply chains. Consumer biomonitoring studies have detected pesticide metabolites in urine and blood that correlate with textile exposure in addition to dietary sources.
Disperse Dyes
Disperse dyes used to color synthetic fibers including polyester, acetate, and nylon are designed to penetrate fiber structures without chemical bonding and this same non-bonding relationship means they migrate freely from fiber to skin surface under conditions of heat and perspiration. Allergic contact dermatitis from disperse dyes is among the most prevalent occupational and consumer skin conditions documented in dermatology literature. The disperse dye compounds most frequently implicated in allergic reactions including Disperse Blue 106, Disperse Blue 124, and Disperse Orange 3 are present in common garment categories including hosiery, sportswear, swimwear, and form-fitting undergarments. Cross-reactivity between disperse dyes and common pharmaceutical compounds including para-aminobenzoic acid sunscreens and certain antibiotics means that individuals sensitized through clothing exposure may develop reactions to unrelated products. The relationship between synthetic garment wear and the rising prevalence of contact dermatitis in the general population is documented in dermatological epidemiology but rarely communicated in retail environments.
Heavy Metal Mordants

Metal-based mordants including chromium, copper, cobalt, nickel, and cadmium compounds are used in textile dyeing to fix colorants to fiber and remain present in finished garments at concentrations that transfer to skin through perspiration-mediated leaching. Nickel is the most common contact allergen in the industrialized world and its presence in textile dyes and finishing agents contributes to sensitization and ongoing reaction in the estimated fifteen to twenty percent of the population carrying nickel hypersensitivity. Chromium compounds used as mordants include hexavalent chromium a compound classified as a known human carcinogen that is restricted in leather processing under European regulations but less consistently controlled in textile dyeing applications. Cadmium accumulates in kidney tissue with a biological half-life measured in decades and its presence in skin-contact textiles represents a chronic low-level exposure pathway that contributes to total body burden alongside dietary and environmental sources. The concentrations of heavy metals in individual garments vary enormously by manufacturer, dye process, and country of origin creating a risk landscape that is invisible to the consumer at the point of purchase.
Optical Brighteners

Optical brightening agents applied to white and light-colored textiles to achieve the luminous whiteness that consumers have been conditioned to associate with cleanliness and quality are stilbene and coumarin derivatives that absorb ultraviolet light and re-emit it as visible blue-white light. These compounds are not chemically bound to textile fibers and transfer to skin surface on contact where they absorb solar ultraviolet radiation against the skin. The photoreactive behavior of optical brighteners on skin produces phototoxic and photoallergic reactions in sensitive individuals upon sun exposure and has been documented as a cause of persistent and treatment-resistant dermatitis in patients whose textile exposure is not initially identified as the causative factor. Stilbene-derived optical brighteners have demonstrated estrogenic activity in cell culture research raising questions about their endocrine activity at skin-contact concentrations. The compounds are also present in laundry detergents and their cumulative application to textiles through repeated washing maintains and in some cases increases their concentration in worn garments over time.
Flame Retardants

Chemical flame retardants applied to children’s sleepwear, upholstered furniture-contact garments, and certain occupational clothing categories include organophosphate compounds, brominated compounds, and antimony trioxide that transfer to skin through direct contact and are detectable in house dust, urine, and breast milk samples from populations with high textile exposure. The regulatory requirement for flame resistance in children’s sleepwear in multiple markets has historically been met through chemical application rather than inherent fiber selection creating a situation in which a safety regulation generates a chemical exposure. Organophosphate flame retardants are structurally related to organophosphate pesticides and share the acetylcholinesterase-inhibiting mechanism that underlies the neurotoxicity of that compound class. Brominated flame retardants have been associated with thyroid disruption, developmental neurotoxicity, and carcinogenicity in experimental research and several compounds have been phased out of use under international agreements while being replaced by structurally similar alternatives whose safety profiles have not been fully characterized. The flame retardant content of garments is not disclosed on product labeling in any major market.
Phthalate Plasticizers

Phthalate compounds used as plasticizers in polyvinyl chloride screen prints, heat transfer labels, rubber appliqués, and synthetic leather components of garments are not chemically bound to the polymer matrices they plasticize and migrate continuously from these components to skin surface through direct contact. Phthalates are classified as endocrine disruptors with specific evidence for anti-androgenic effects that have been associated with reproductive development outcomes in studies of prenatal and postnatal exposure. The migration rate of phthalates from garment components accelerates with heat and friction making activewear and garments worn during physical activity particularly effective delivery vehicles for these compounds to the skin and bloodstream. Di-2-ethylhexyl phthalate and dibutyl phthalate are restricted in children’s products in multiple jurisdictions but appear in adult garments and accessories without equivalent regulation. Urinary phthalate metabolite concentrations in the general population are elevated in individuals with higher synthetic garment and accessory contact and the textile exposure pathway contributes meaningfully to total body burden alongside food packaging and cosmetic sources.
Chlorine Bleaching

Chlorine-based bleaching agents used in textile processing to achieve uniform whiteness in natural fibers generate organochlorine compounds as reaction byproducts that persist in finished fabric and transfer to skin through contact and perspiration. Dioxins and furans produced as chlorine bleaching byproducts are among the most toxic compounds produced by industrial processes and their presence in chlorine-bleached textiles at trace levels represents a chronic low-level exposure pathway in the population. The environmental persistence and bioaccumulation potential of organochlorine bleaching byproducts means that their presence in textiles contributes to a broader body burden that accumulates alongside dietary and environmental exposures across a lifetime. Hydrogen peroxide and ozone bleaching processes produce equivalent whitening results without the organochlorine byproduct profile but adoption of these alternatives remains incomplete across the global textile supply chain. Certified organic textile standards typically require chlorine-free bleaching but this information is not communicated through mainstream retail labeling.
Nonylphenol Ethoxylates

Nonylphenol ethoxylates used as surfactants and emulsifiers in textile dyeing and processing are not fully removed during finishing and persist in garments at concentrations that have been detected in human urine following skin contact during wear. These compounds degrade in biological and environmental systems to nonylphenol a potent xenoestrogen that mimics estrogen activity at concentrations significantly lower than those associated with acute toxicity. Greenpeace’s Detox campaign testing of major fashion brand products from multiple countries consistently detected nonylphenol ethoxylate residues across brand tiers from mass market to premium. The compounds are restricted in textile processing within the European Union but their use persists in manufacturing regions supplying global markets without equivalent regulatory frameworks. The estrogenic activity of nonylphenol at skin-contact concentrations is a subject of active regulatory and academic debate with the textile exposure pathway representing an undercharacterized contribution to total xenoestrogen body burden.
Antimicrobial Treatments

Silver nanoparticles, triclosan, and quaternary ammonium compounds applied to sportswear, socks, and odor-resistant clothing as antimicrobial finishes transfer to skin during wear and disrupt the skin microbiome by targeting beneficial microbial populations alongside odor-producing bacteria. The skin microbiome performs critical functions in barrier maintenance, immune education, and pathogen resistance and its disruption through chronic antimicrobial exposure has been associated with increased rates of skin infection, atopic dermatitis, and allergic sensitization in research populations. Triclosan specifically has been associated with antibiotic resistance development, thyroid hormone disruption, and skin sensitization and has been removed from personal care products in several major markets while remaining in textile applications without equivalent regulatory attention. Silver nanoparticles released from treated textiles during wear and washing accumulate in biological tissue and have demonstrated cytotoxicity and genotoxicity in cell culture studies at concentrations achievable through chronic dermal exposure. The antimicrobial function of these treatments diminishes with washing while the sensitization and microbiome disruption effects accumulate with continued exposure.
Synthetic Fragrance

Fragrance compounds applied to clothing through manufacturing processes, retail environment spraying, and scented packaging include hundreds of individual chemical constituents whose identities are protected as proprietary formulas and are therefore not disclosed on any product labeling. The fragrance compounds that most consistently produce dermatological and systemic effects through textile contact include musks, benzophenones, and specific aldehyde and terpene derivatives that are absorbed transdermally and detected in human blood, urine, and breast milk. Nitromusks and polycyclic musks used in textile fragrance applications have demonstrated endocrine activity and bioaccumulation potential and several compounds in these classes have been restricted in the European Union. The cumulative fragrance exposure from scented clothing worn against skin for twelve or more hours daily represents a chronic low-level chemical exposure pathway that is distinct from and additional to cosmetic and household product fragrance sources. Individuals who develop fragrance sensitivity through cumulative exposure may experience cross-reactive responses to unrelated products that were previously well tolerated.
Polyester Microplastics

The mechanical stress of wearing polyester and other synthetic fiber garments releases microplastic particles from fiber surfaces that deposit on skin and in hair follicles where they act as carriers for the chemical additives incorporated during fiber manufacturing including flame retardants, plasticizers, and dye compounds. The skin deposition of microplastics from worn garments occurs continuously throughout the wearing day and the concentration of particles on skin surface increases with garment age, wear frequency, and physical activity level. Synthetic fiber microplastics have been detected in human blood, lung tissue, placental tissue, and stool samples in recent biomonitoring research confirming systemic uptake through multiple routes including dermal absorption. The chemical payloads carried by microplastic particles include compounds that would not penetrate intact skin independently but that are delivered to skin surface and follicular structures by the particles that carry them. The total dermal microplastic exposure from synthetic garment wear has not been fully characterized but represents a newly recognized exposure pathway with implications extending beyond the skin to systemic tissue distribution.
Rubber Elastics

Rubber components including elastic waistbands, bra underwires encased in rubber, and elastic cuffs contain latex proteins, mercaptobenzothiazole, thiuram compounds, and carbamate accelerators used in rubber vulcanization that are among the most potent contact allergens identified in dermatological research. Latex allergy affects an estimated one to six percent of the general population and a significantly higher proportion of individuals with repeated rubber exposure, and reactions range from localized contact dermatitis to systemic anaphylaxis in severely sensitized individuals. The rubber accelerator compounds mercaptobenzothiazole and thiuram are ranked among the top ten most common causes of allergic contact dermatitis in patch test studies across multiple countries. The contact between rubber-containing garment components and skin occurs at high-pressure points including waistbands and bra bands where the combination of pressure, heat, and perspiration maximizes transfer of allergenic compounds. The prevalence of rubber component allergies in the population is substantially underestimated because many affected individuals attribute their reactions to soap, detergent, or food rather than to the clothing component against which the reaction consistently localizes.
Chromium Leather Accessories

Chrome-tanned leather used in belts, watchstraps, shoe linings, and bag contact surfaces that rest against skin undergoes ongoing oxidation that converts residual trivalent chromium in the leather to hexavalent chromium, a known human carcinogen and potent contact allergen. The conversion occurs more rapidly in conditions of heat, perspiration, and ultraviolet light exposure all of which are present in the typical use environment of leather accessories. Hexavalent chromium contact dermatitis from leather goods is a well-documented clinical entity with a globally increasing prevalence that dermatologists attribute in part to the growing volume of chrome-tanned leather products in consumer use. European Union regulation restricts hexavalent chromium in leather products to concentrations below three parts per million but enforcement testing of products at retail has repeatedly identified non-compliant items at concentrations substantially above this threshold. Individuals who develop chromium sensitivity through leather contact become reactive to chromium in unrelated products including certain cements, paints, and metal alloys.
Wrinkle Resistant Shirts

The specific chemistry of wrinkle-resistant dress shirt treatments deserves separate examination from general formaldehyde finishing because these garments are engineered for sustained professional wear in conditions of physical warmth and perspiration that maximize transdermal transfer of finishing agents. Dimethylol dihydroxyethylene urea and related N-methylol compounds used in these treatments release formaldehyde continuously throughout the garment’s useful life and the release rate increases with body heat. The collar and cuff areas of dress shirts concentrate formaldehyde exposure at locations directly adjacent to facial skin and pulse points where skin is thinner and more vascular than elsewhere on the body. Occupational dermatitis studies have consistently identified dress shirts and other professional wrinkle-resistant garments as causal agents in contact dermatitis cases presenting in working populations. The marketing of these garments emphasizes convenience and professional appearance without any disclosure of the chemical engineering that produces the performance benefit.
Dark Denim

Dark and black denim achieves its depth of color through the application of synthetic indigo, sulfur dyes, and finishing chemicals in concentrations substantially higher than those required for lighter colorways and these compounds transfer to skin at measurable rates throughout the garment’s useful life. The sulfur dye compounds used in dark denim finishing include thiosulfate derivatives that produce hydrogen sulfide as a degradation product, a compound that contributes to skin irritation in individuals with high denim contact. Para-phenylenediamine used in some dark textile dyeing applications is one of the most potent contact allergens in consumer use and a sensitizer that creates permanent hypersensitivity capable of producing reactions to structurally related compounds including certain hair dyes and sunscreen chemicals. The dye transfer from dark denim to skin is visible in individuals who wear new dark jeans against pale skin and the visible transfer is accompanied by invisible transfer of reactive dye constituents. Washing dark denim before first wear reduces but does not eliminate the initial dye transfer and the ongoing migration of dye compounds from fiber to skin continues throughout the garment’s lifespan.
Nanosilver Technology

Nanosilver particles engineered at scales between one and one hundred nanometers and incorporated into fabric structures at the fiber level rather than applied as surface coatings represent a more biologically available form of silver than conventional antimicrobial treatments because nanoparticle size enables penetration of skin barrier structures that larger particles cannot traverse. The cellular toxicity of nanosilver has been demonstrated in multiple tissue and cell culture systems at concentrations achievable through chronic dermal contact with treated garments. Nanosilver particles interact with skin proteins to generate reactive oxygen species that produce oxidative damage to cellular structures at the skin surface and in deeper dermal layers accessed through follicular penetration pathways. The environmental release of nanosilver through garment washing contributes to aquatic ecosystem disruption but the dermal exposure of garment wearers represents a more direct and concentrated exposure pathway. Disclosure of nanoparticle use in textile products is not required in most major markets and the word silver in performance garment marketing encompasses both conventional and nano-scale silver treatments without distinction.
Textile Softeners

Industrial fabric softeners applied during finishing to improve the hand feel and drape of textiles include quaternary ammonium compounds, silicone derivatives, and polyethylene wax emulsions that coat fiber surfaces and transfer to skin through contact, producing biological effects that range from simple barrier disruption to documented sensitization and hormonal activity. Dimethyl dialkyl ammonium chloride and related quaternary ammonium fabric softener compounds are biocidal agents that disrupt cellular membrane integrity and have demonstrated cytotoxicity in skin cell models at concentrations comparable to those present in treated garments. The silicone compounds used in textile softening accumulate in the skin’s lipid barrier and alter its permeability in ways that can both reduce and increase the absorption of other compounds depending on the specific silicone structure. Consumer fabric softeners applied during home laundering add an additional layer of these compounds on top of the industrial softening treatment applied during manufacturing. The cumulative softener load on regularly laundered garments represents an ongoing skin contact chemical exposure that is entirely unregulated and undisclosed.
Reactive Dyes
Reactive dyes used to color cellulosic fibers including cotton, linen, and viscose form covalent bonds with fiber hydroxyl groups but the bonding reaction is never complete and the unfixed dye fraction that remains in finished garments transfers to skin through hydrolysis in the presence of perspiration. The hydrolyzed reactive dye compounds are more water-soluble and therefore more dermally available than their fiber-bonded counterparts and include structures that have demonstrated mutagenic activity in bacterial assay systems. The vinylsulfone and dichlorotriazine reactive groups present in many commercially important reactive dyes are chemically reactive at physiological conditions and produce protein-haptenation reactions with skin proteins that underlie their capacity to induce allergic sensitization. Reactive dye sensitization, once established, produces responses to structurally related compounds in products far removed from textiles including certain medications, food colorants, and industrial chemicals encountered in occupational settings. The fiber-bonded fraction of reactive dyes is considered safe but the unfixed fraction present in commercially produced garments is a function of the dyeing process efficiency which varies substantially between manufacturers.
Residual Solvents

Organic solvents including dimethylformamide, toluene, benzene derivatives, and chlorinated hydrocarbons used in textile printing, coating, and adhesive applications remain in finished garments as residual contaminants whose volatilization against warm skin provides a continuous low-level inhalation and dermal exposure pathway. Dimethylformamide specifically used in polyurethane coating applications for faux leather and technical textile surfaces is a reproductive toxin and liver toxin with a well-documented occupational hazard profile that has received significantly less attention in the context of consumer garment contact. The residual solvent burden in a new garment is highest immediately after manufacture and diminishes with airing and washing but the off-gassing period during which skin contact occurs at its most chemically productive is precisely the initial wearing period before the consumer has established a laundering pattern. Enclosed environments including cars and offices where a new garment is worn accumulate volatile compounds to concentrations that can be measured by air quality monitoring equipment. The characteristic smell of new clothing, particularly items containing synthetic coatings and prints, is olfactory evidence of the volatile solvent residues that chemical analysis confirms are present.
Sizing Agents

Textile sizing compounds applied to yarns and fabrics during weaving to provide rigidity and surface smoothness for manufacturing processing include polyvinyl alcohol, modified starches, polyacrylate derivatives, and carboxymethyl cellulose formulations that remain in fabric after processing and are not fully removed during conventional consumer laundering. Polyacrylate sizing compounds have demonstrated sensitizing potential and their presence in skin-contact textiles contributes to the background rate of contact dermatitis associated with new garment wear. The sizing load on a new garment stiffens the fabric in ways that increase mechanical friction against skin producing microabrasion that elevates the permeability of the skin barrier to the chemical compounds simultaneously being transferred from the garment. The combination of sizing-induced barrier disruption and concurrent chemical transfer represents a synergistic exposure mechanism that is greater in impact than either factor independently. Washing new garments before first wear removes a substantial proportion of sizing compounds and is consistently recommended in textile chemistry literature though this recommendation is communicated to consumers inconsistently and without explanation of the specific chemical rationale.
Polyurethane Coatings

Polyurethane coatings applied to faux leather garments, coated denim, water-resistant jackets, and technical outerwear contain residual isocyanate compounds from incomplete polymerization reactions that are among the most potent respiratory and dermal sensitizers encountered in occupational chemical exposure settings. Diisocyanate compounds represent the leading cause of occupational asthma in manufacturing environments and their presence as residuals in consumer garments creates a dermal and inhalation exposure pathway for sensitization in the general population. The isocyanate content of polyurethane-coated garments off-gases more rapidly in conditions of warmth and wear and the body heat generated under a close-fitting coated garment creates a microenvironment that accelerates this release against skin. Once isocyanate sensitization is established through any exposure route the individual becomes permanently reactive to these compounds at extremely low concentrations making subsequent exposures in occupational or consumer contexts capable of triggering severe responses. The coated garment category has expanded dramatically with the growth of faux leather fashion and the concurrent isocyanate exposure profile of general consumer populations has not been systematically characterized.
Chlorinated Pool Interactions

Chlorine-resistant swimwear manufactured to maintain color and structural integrity under pool chemical conditions contains fixatives, UV stabilizers, and elastane treatments whose chemical stability under chlorine exposure releases reaction products at the skin contact surface during aquatic use. The combination of pool chlorine, body perspiration, and swimwear chemical residues at the skin interface creates a reactive chemical environment that is distinctly more complex than any individual component. Disinfection byproducts including chloramines formed through the reaction of pool chlorine with organic compounds from garment finishes and body fluids produce skin and mucous membrane irritation that most swimmers attribute entirely to the pool water rather than the garment contribution. The skin barrier disruption produced by extended aquatic exposure maximizes dermal absorption of whatever compounds are present at the skin surface making swimwear chemical residues more bioavailable during pool use than the same compounds would be during dry wear conditions. The intersection of garment chemistry and pool chemistry as a combined skin exposure environment is an area where consumer awareness is essentially zero.
Fast Fashion Finishing

The compressed manufacturing timelines characteristic of fast fashion production result in garments where chemical treatment processes including dyeing, finishing, and coating are insufficiently cured, washed, or processed before the garment is packaged and shipped to retail. Insufficient post-dyeing washing leaves substantially higher concentrations of unfixed dye, processing chemicals, and reactive intermediates in the finished garment than properly processed equivalents. The economic pressure on fast fashion supply chains to reduce water and energy use in processing exacerbates this problem as thorough washing and curing of chemical treatments is resource-intensive. Independent testing of fast fashion garments consistently identifies chemical residue levels at the higher end of the distribution across all categories of textile chemical contamination. The relationship between garment price point and chemical residue burden is not perfectly linear but the systemic pressures operating on ultra-low-cost manufacturing create conditions in which thorough chemical processing is structurally disincentivized.
Bamboo Processing

Bamboo fabric marketed as a natural and sustainable alternative to conventional textiles is in the majority of commercial applications a regenerated cellulose fiber produced through the viscose process using sodium hydroxide and carbon disulfide solvents that convert bamboo pulp into a fiber with no structural relationship to the original plant. Carbon disulfide is a neurotoxin with a well-documented occupational hazard profile in viscose production workers and its residual presence in viscose bamboo fabric at concentrations that transfer to skin during wear represents a consumer exposure pathway that is entirely obscured by the natural and eco-friendly marketing narrative surrounding these products. The sustainable framing of bamboo textiles creates an impression of chemical simplicity and environmental benignity that is directly contradicted by the chemistry of the production process. Bamboo viscose garments carry essentially the same chemical residue profile as conventional viscose and rayon products processed through identical manufacturing methods. The Federal Trade Commission has taken enforcement action against companies making unsubstantiated natural and organic claims for viscose bamboo products in the United States market.
Rental and Secondhand Chemicals

Garments circulated through clothing rental services and processed through commercial cleaning operations are treated with industrial-grade cleaning solvents, disinfectants, and finishing agents between uses that differ substantially in their chemical profiles from consumer laundering products and are applied at concentrations calibrated for fiber performance rather than skin contact safety. Perchloroethylene the dominant dry cleaning solvent is a probable human carcinogen and central nervous system toxin whose residual presence in dry-cleaned garments is detectable by sensitive individuals through odor and is measurable analytically at concentrations that transfer to skin during wear. The commercial finishing sprays applied after dry cleaning to restore hand feel and appearance include acrylate polymers, silicone compounds, and fragrance formulations that add a chemical layer above the base textile chemistry. Rental garments worn against skin by sequential users carry a cumulative chemical history that reflects the cleaning and treatment practices of the rental operator rather than the manufacturing chemistry of the original garment. The assumption that commercial cleaning removes rather than adds chemical complexity to garment surfaces is a consumer misconception that the chemistry of commercial textile care does not support.
Waistband Rubber

The specific contact zone of trouser and skirt waistbands against the skin of the lower torso deserves individual examination because this area combines high contact pressure, ambient warmth from the body’s core, and typically higher perspiration levels than extremities creating a chemical transfer environment that is more biologically productive than most garment contact zones. Elastic waistbands containing rubber accelerator compounds maintain sustained pressure contact with skin in a zone where the combination of temperature and moisture maximizes allergen transfer and dermal absorption. The prevalence of waistband contact dermatitis presenting as a band of inflammation precisely following the waistband perimeter is a well-documented dermatological pattern that most affected individuals spend extended periods misattributing to soap, food, or environmental allergens. Nickel-containing metal waistband hardware combined with rubber elastic compounds creates a multi-allergen contact zone at a single anatomical location. Dermatologists report that waistband-localized dermatitis that clears with removal of the specific garment and recurs with its replacement is among the most diagnostically straightforward but practically underrecognized patterns in contact dermatitis clinical practice.
Antistatic Treatments

Antistatic compounds applied to synthetic fiber garments to reduce the static electricity buildup that produces discomfort and garment cling include quaternary ammonium salts, phosphate esters, and ethoxylated fatty acid derivatives that coat fiber surfaces and transfer continuously to skin during wear. The antistatic function of these compounds is directly dependent on their ability to migrate to the fiber surface and attract atmospheric moisture and this same surface migration behavior governs their transfer to skin in contact with treated fabric. Quaternary ammonium antistatic compounds share structural and functional properties with the cationic surfactants used as fabric softeners and disinfectants and their cytotoxic and sensitizing potential is similarly documented. Phosphate ester antistatic agents are metabolized to bioactive phosphate derivatives in biological systems and their endocrine activity has been investigated in the context of their widespread use as flame retardants in addition to their antistatic applications. The chemical overlap between antistatic, softening, and antimicrobial textile treatments means that the compounds a consumer encounters through garment contact frequently belong to multiple functional categories simultaneously.
UV Stabilizers

Ultraviolet light stabilizers incorporated into synthetic fibers and applied to fabric surfaces to prevent photodegradation of dye compounds and fiber polymers include benzophenone derivatives and benzotriazole compounds that transfer to skin and interact with solar ultraviolet radiation in ways that can paradoxically increase rather than decrease skin photochemical stress. Benzophenone-3 and related benzophenone UV stabilizers are classified as endocrine disruptors with estrogenic and androgenic activity at concentrations detected in human urine from populations with significant synthetic textile contact. The photoreactivity of benzophenone compounds on skin produces reactive oxygen species and protein adducts that represent a photochemical skin exposure pathway distinct from direct solar UV exposure. Benzophenone UV stabilizers in textiles are distinct from but structurally related to benzophenone UV filters in sunscreens and the sensitization produced through garment contact can generate cross-reactive responses to sunscreen products. The presence of UV stabilizers in textile chemistry is essentially invisible to consumers and is not addressed by any existing garment labeling requirement.
Chlorobenzene Carriers

Carrier compounds used in polyester dyeing to swell fiber structures and allow dye penetration at lower temperatures include chlorinated benzene derivatives and biphenyl compounds that remain in fiber after dyeing as residual contaminants with documented neurotoxic and hepatotoxic profiles. The carrier dyeing process is used extensively in polyester production because it reduces energy costs relative to high-temperature dyeing methods and the economic advantages of the process have sustained its use despite the toxicological profiles of the carrier compounds employed. Residual carrier compounds in polyester garments transfer to skin in conditions of body heat and perspiration and the lipophilic nature of these compounds supports dermal absorption and systemic distribution. The hepatotoxic potential of chlorinated carrier residues is relevant given that chronic low-level liver toxin exposure through dermal routes is less studied than acute high-level occupational exposure but may produce subclinical effects detectable only in long-term biomonitoring. Independent analysis of polyester garments from mainstream retail sources has detected carrier compound residues at concentrations varying widely between manufacturers reflecting the significant process control differences in the global polyester dyeing industry.
Mordant Metals in Prints

Screen printing inks and heat transfer print systems applied to garment surfaces contain metallic compounds used as pigment components, binding agents, and adhesion promoters including chromium, lead, cadmium, and cobalt that transfer to skin from print surfaces through the mechanical action of wear. Print areas on garments maintain sustained contact with skin under pressure and heat and the compound transfer from these areas is concentrated relative to the transfer from dyed ground fabric. Lead compounds historically used as stabilizers in PVC plastisol screen printing inks are subject to restriction in children’s products in multiple jurisdictions but appear in adult garments and in products manufactured outside regulated markets at concentrations substantially above acceptable thresholds. Cadmium-based yellow and orange pigments produce the specific hues most difficult to achieve through cadmium-free chemistry and their replacement with safer alternatives has progressed unevenly across the global garment printing industry. The print areas of heavily decorated garments represent localized high-intensity chemical transfer zones that are distinct from and additional to the chemical exposure profile of the garment’s base fabric.
Recycled Fiber Contamination

Garments manufactured from recycled synthetic fibers including post-consumer recycled polyester carry chemical contaminants from the previous use of the source materials including food contact compounds, packaging chemicals, industrial processing residues, and cleaning product accumulations that persist through mechanical recycling processes that do not include chemical purification steps. The recycled content designation on garments communicates an environmental benefit while providing no information about the chemical history of the source material or the residual contaminant profile of the fiber produced. Antimony trioxide used as a catalyst in virgin polyester production and present as a residual in recycled polyester fibers is a suspected human carcinogen and skin irritant that transfers to skin during wear at rates that are a function of body temperature and perspiration levels. The chemical complexity of recycled fiber garments is potentially higher than that of virgin fiber equivalents precisely because recycling processes consolidate the chemical histories of diverse source streams into a single fiber product. Consumer demand for recycled content garments as an environmental choice has grown substantially faster than the analytical characterization of the chemical implications of recycled fiber use in skin-contact textile applications.
Waterproofing Sprays

Aftermarket waterproofing and fabric protection sprays applied to garments by consumers at home contain fluoropolymer compounds, acrylate polymers, and solvent carriers that deposit a chemical treatment layer on fabric surfaces that was not present in the original garment’s manufacturing chemistry. The consumer application of these products in enclosed domestic spaces creates acute inhalation exposure risks that have been associated with pulmonary toxicity events in documented case reports. The fluoropolymer compounds in aftermarket waterproofing sprays belong to the same PFAS chemical family as the manufacturing-applied performance finishes described earlier and their home application adds a fresh treatment layer that may deliver higher initial surface concentrations than aged factory-applied treatments. The solvent carriers in aerosol waterproofing formulations include compounds that penetrate fabric and deposit the active compounds at fiber surfaces throughout the fabric thickness rather than solely on the outer surface, maximizing contact between the treatment and the skin-facing inner surface of the garment. The home application of these products is essentially unregulated from a consumer product safety standpoint and the labeling information provided on most products is insufficient to communicate the nature and duration of chemical exposure they create during and following application.
Interactive Sweat Chemistry

The chemistry of perspiration itself transforms the hazard profile of multiple textile chemical categories by acting as an extraction medium that mobilizes compounds from fiber surfaces and as a reactive medium that chemically modifies textile compounds into derivatives with different and sometimes greater biological activity. The organic acids, enzymes, and urea compounds present in perspiration create conditions at the skin-fabric interface that are chemically productive in ways that laboratory testing of dry garments does not capture. Alkaline perspiration specifically mobilizes metal compounds from fiber surfaces at rates substantially higher than those measured in neutral aqueous extraction testing used for compliance assessment, meaning that regulatory testing may significantly underestimate real-world metal transfer. The metabolic transformation of azo dye compounds to carcinogenic amines occurs through bacterial reduction and the bacterial populations present in perspiration-wetted fabric are more metabolically active than those present in standard test conditions. The garment worn during exercise in conditions of heavy perspiration and elevated skin temperature creates a chemical transfer environment that represents the worst case for virtually every category of textile chemical contaminant and this worst-case scenario is precisely the condition targeted by performance and activewear marketing.
Supply Chain Laundering

The multi-stage global supply chain through which most garments pass before reaching the consumer generates chemical additions at each processing stage that are invisible to the end consumer and undisclosed in any required labeling format. A single garment may have its fiber grown in one country, spun in a second, woven in a third, dyed and finished in a fourth, cut and assembled in a fifth, and labeled and distributed through a sixth, with each transition representing a potential addition of processing chemicals, transport treatments, and storage compounds. Anti-mold agents applied to garments for long-distance maritime shipping include isothiazolinone compounds and thiocyanate derivatives that are potent contact allergens and that are not required to be declared on any consumer-facing product information. The consolidation of chemical additions across a six to ten stage supply chain produces a final product whose chemical complexity cannot be inferred from brand reputation, price point, or country of final assembly labeling. Independent textile safety certification programs including OEKO-TEX and GOTS represent the most practically accessible mechanism for consumers to reduce supply chain chemical uncertainty but their adoption remains voluntary and their premium positioning limits their reach in mass market retail.
Hormone Disruption Synergy

The collective endocrine-disrupting potential of the chemical compounds present in a typical garment wardrobe exceeds the sum of the individual contributions of each compound because endocrine disruptors operating through the same hormonal pathway produce additive and sometimes synergistic effects at combined concentrations below the threshold at which any individual compound would produce a measurable effect alone. This mixture toxicity principle means that regulatory assessment of individual textile chemicals at individual compound concentrations systematically underestimates the real-world endocrine disruption potential of the chemical exposure produced by wearing multiple garments with multiple low-level endocrine-active compounds simultaneously. The estrogenic activity of textile chemical mixtures including nonylphenol ethoxylates, benzophenone UV stabilizers, phthalate plasticizers, and phytoestrogenic dye compounds is greater in combination than compound-by-compound risk assessment frameworks capture. The same mixture principle applies to the combined neurotoxic, hepatotoxic, and carcinogenic potential of co-occurring textile chemical residues and represents the most fundamental limitation of the regulatory frameworks through which textile chemical safety is currently assessed. Consumer exposure to the full mixture of textile chemicals present in a daily wardrobe has not been systematically characterized by any regulatory body and the endocrine and systemic health implications of this mixture exposure across a lifetime remain one of the most significant and least addressed questions in environmental health science.
If you have noticed unexplained skin reactions or sensitivities that you suspect may be connected to the clothing you wear share your experiences and discoveries in the comments.